stdClass Object
(
[nazev] => Central Laboratories UCT Prague
[adresa_url] =>
[api_hash] =>
[seo_desc] =>
[jazyk] =>
[jednojazycny] =>
[barva] =>
[indexace] => 1
[obrazek] =>
[ga_force] =>
[cookie_force] =>
[secureredirect] =>
[google_verification] =>
[ga_account] =>
[ga_domain] =>
[ga4_account] => G-VKDBFLKL51
[gtm_id] =>
[gt_code] =>
[kontrola_pred] =>
[omezeni] =>
[pozadi1] =>
[pozadi2] =>
[pozadi3] =>
[pozadi4] =>
[pozadi5] =>
[robots] =>
[htmlheaders] =>
[newurl_domain] => 'clab.vscht.cz'
[newurl_jazyk] => 'en'
[newurl_akce] => '[en]'
[newurl_iduzel] =>
[newurl_path] => 1/20076/20078
[newurl_path_link] => Odkaz na newurlCMS
[iduzel] => 20078
[platne_od] => 26.05.2023 09:28:00
[zmeneno_cas] => 26.05.2023 09:28:48.557372
[zmeneno_uzivatel_jmeno] => Jan Kříž
[canonical_url] =>
[idvazba] => 24782
[cms_time] => 1713610007
[skupina_www] => Array
(
)
[slovnik] => stdClass Object
(
[logo_href] => /
[logo] =>  [logo_mobile_href] => /
[logo_mobile] =>
[logo_mobile_href] => /
[logo_mobile] =>  [google_search] => 001523547858480163194:u-cbn29rzve
[social_fb_odkaz] =>
[social_tw_odkaz] =>
[social_yt_odkaz] =>
[intranet_odkaz] => http://intranet.vscht.cz/
[intranet_text] => Intranet
[mobile_over_nadpis_menu] => Menu
[mobile_over_nadpis_search] => Search
[mobile_over_nadpis_jazyky] => Languages
[mobile_over_nadpis_login] => Login
[menu_home] => Homepage
[paticka_budova_a_nadpis] => BUILDING A
[paticka_budova_a_popis] => Rector,
Department of Communications,
Department of Education,
FCT Dean’s Office,
Centre for Information Services
[paticka_budova_b_nadpis] => BUILDING B
[paticka_budova_b_popis] =>
Department of R&D, Dean’s Offices:
FET,
FFBT,
FCE,
Computer Centre,
Department of International Relations,
Bursar
[paticka_budova_c_nadpis] => BUILDING C
[paticka_budova_c_popis] => Crèche Zkumavka,
General Practitioner,
Department of Economics and Management,
Department of Mathematics
[paticka_budova_1_nadpis] => NATIONAL LIBRARY OF TECHNOLOGY
[paticka_budova_1_popis] =>
[paticka_budova_2_nadpis] => CAFÉ CARBON
[paticka_budova_2_popis] =>
[paticka_adresa] => UCT Prague
[google_search] => 001523547858480163194:u-cbn29rzve
[social_fb_odkaz] =>
[social_tw_odkaz] =>
[social_yt_odkaz] =>
[intranet_odkaz] => http://intranet.vscht.cz/
[intranet_text] => Intranet
[mobile_over_nadpis_menu] => Menu
[mobile_over_nadpis_search] => Search
[mobile_over_nadpis_jazyky] => Languages
[mobile_over_nadpis_login] => Login
[menu_home] => Homepage
[paticka_budova_a_nadpis] => BUILDING A
[paticka_budova_a_popis] => Rector,
Department of Communications,
Department of Education,
FCT Dean’s Office,
Centre for Information Services
[paticka_budova_b_nadpis] => BUILDING B
[paticka_budova_b_popis] =>
Department of R&D, Dean’s Offices:
FET,
FFBT,
FCE,
Computer Centre,
Department of International Relations,
Bursar
[paticka_budova_c_nadpis] => BUILDING C
[paticka_budova_c_popis] => Crèche Zkumavka,
General Practitioner,
Department of Economics and Management,
Department of Mathematics
[paticka_budova_1_nadpis] => NATIONAL LIBRARY OF TECHNOLOGY
[paticka_budova_1_popis] =>
[paticka_budova_2_nadpis] => CAFÉ CARBON
[paticka_budova_2_popis] =>
[paticka_adresa] => UCT Prague
Technická 5
166 28 Prague 6 – Dejvice
IČO: 60461373 / VAT: CZ60461373
Czech Post certified digital mail code: sp4j9ch
Copyright: UCT Prague 2015
Information provided by the Department of International Relations and the Department of R&D. Technical support by the Computing Centre.
[paticka_odkaz_mail] => mailto:Jan.Prchal@vscht.cz
[zobraz_desktop_verzi] => switch to full version
[social_fb_title] =>
[social_tw_title] =>
[social_yt_title] =>
[aktualizovano] => Updated
[autor] => Author
[drobecky] => You are here: UCT Prague – Central Laboratories
[paticka_mapa_odkaz] =>
[zobraz_mobilni_verzi] => switch to mobile version
[nepodporovany_prohlizec] => For full access, please use different browser.
[stahnout] => Download
[preloader] => Wait a second...
[social_in_odkaz] =>
[social_li_odkaz] =>
)
[poduzel] => stdClass Object
(
[20081] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[20093] => stdClass Object
(
[obsah] =>
[iduzel] => 20093
[canonical_url] => //clab.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[20091] => stdClass Object
(
[obsah] =>
[iduzel] => 20091
[canonical_url] => //clab.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[20092] => stdClass Object
(
[obsah] =>
[iduzel] => 20092
[canonical_url] => //clab.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
)
[iduzel] => 20081
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[20082] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[27385] => stdClass Object
(
[nazev] => Laboratory of Atomic Absorption Spectroscopy (AAS)
[seo_title] => Laboratory of Atomic Absorption Spectroscopy (AAS)
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] => Analysis
The Atomic Absorption Spectroscopy (AAS) is used in the laboratory to measure the content of metals in inorganic or organic samples. The AAS can be performed with both flame and electrothermal atomization. The samples are measured in the form of solutions in water or organic phase (methanol, ethanol). In some cases, decomposition methods can be employed. The determination of mercury can be performed also in solid samples.
The following elemental analyses are performed in the laboratory (detection limit in mg/l for flame atomization):
|
Ag (0.03)
|
Al (0.4)
|
As (0.6)
|
Au (0.1)
|
|
|
|
B (8)
|
Ba (0.2)
|
Be (0.02)
|
Bi (0.2)
|
|
|
|
Ca (0.01)
|
Cd (0.005)
|
Co (0.05)
|
Cr (0.06)
|
Cs (0.04)
|
Cu (0.04)
|
|
Fe (0.05)
|
Ga (0.7)
|
Ge (1.5)
|
Hg (5)
|
In (0.2)
|
|
|
K (0.01)
|
La (1)
|
Li (0.02)
|
Mg (0.003)
|
Mn (0.02)
|
Mo (0.3)
|
|
Na (0.003)
|
Ni (0.05)
|
Pb (0.1)
|
Pd (0.1)
|
Pt (1.5)
|
|
|
Rb (0.03)
|
Rh (0.15)
|
Sb (0.3)
|
Se (0.5)
|
Si (1.0)
|
|
|
Sn (1)
|
Sr (0.05)
|
Ta (11)
|
Ti (1.5)
|
|
|
|
Tl (0.3)
|
V (0.75)
|
W (6)
|
Zn (0.05)
|
|
|
For some elements, it is possible to achieve lower detection limits by combination of AAS with electrothermal atomization (Hg) or AAS with hydride technique (As, Se, Sb). These methods have to be discussed individually.
Other activities
- consultancy
- research and monitoring in the field of health care and environment
Environmental protection
Analysis of rain water, underground water, and waste water
Air quality, pollution, aerosol particles
Research in the field of population health and environment (government project)
Preparation of tested samples, testing of sample collection machines, research in the field of elemental analysis in aerosol particles (As, Cd, Cr, Mn, Ni, Pb)
[iduzel] => 27385
[canonical_url] => //clab.vscht.cz/aas-en
[skupina_www] => Array
(
)
[url] => /aas-en
[sablona] => stdClass Object
(
[class] => stranka_submenu
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27703] => stdClass Object
(
[nazev] => Laboratory of NMR Spectroscopy
[seo_title] => Laboratory of NMR Spectroscopy
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
Laboratory of NMR spectroscopy is the part of the Central Laboratories of the University of Chemistry and Technology (ICT) in Prague. The main goal of our laboratory is to measure service NMR samples delivered not only by various school laboratories but also by out-of-school institutions. Apart from basic services we are able to perform more complex non-standard experiments involving their interpretation and evaluation. At the same time our laboratory deals with scientific and teaching activities that involve close cooperation with other departments of ICT.
NMR spectroscopy can be widely used for characterization of organic compounds. In addition to determination of chemical structure of various products and reactions intermediates, NMR is applicable also for mixture component analysis, kinetic and thermodynamic measurements. NMR is also a powerful tool for quantitative and qualitative calculation of geometry of the molecules and their dynamics.
Enormous development of advanced techniques enabled NMR spectroscopy to become the significant method for calculation of the structure of biomacromolecules – proteins and nucleic acids. At present we are solving the spatial geometry of selected proteins of Mason-Pfizer monkey virus.
[iduzel] => 27703
[canonical_url] => //clab.vscht.cz/nmr_en
[skupina_www] => Array
(
)
[url] => /nmr_en
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27387] => stdClass Object
(
[nazev] => Laboratory of transmission electron microscopy
[seo_title] => Laboratory of transmission electron microscopy
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
page under construction
[iduzel] => 27387
[canonical_url] => //clab.vscht.cz/tem-en
[skupina_www] => Array
(
)
[url] => /tem-en
[sablona] => stdClass Object
(
[class] => stranka_submenu
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[21615] => stdClass Object
(
[nazev] => Central Laboratories
[seo_title] => UCT Prague - Central Laboratories
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
The main goal of Central Laboratories at the University of Chemistry and Technology, Prague is to measure service samples delivered by various school laboratories. The analytical services are used for grant and students projects, industrial research application. Analyses are also offered to external customers (universities, private companies etc.). Apart from basic routine analyses more complex non-standard experiments are also performed. The laboratories are equipped with a modern and top-level instrumentation and the the laboratory staff deals with scientific and teaching activities that involve close cooperation with the departments of the University. Central Laboratories consist of nine laboratories, detailed information can be found on their pages.
Richard Hrabal, associate professor
Head of Central Laboratories
| Laboratory of Nuclear Magnetic Resonance (NMR) Spectroscopy |
Head: Richard Hrabal (richard.hrabal@vscht.cz)
Phone: +420 220 443 805
building A, room 42, ground-floor |
| Laboratory of Infra-Red (IR) and Raman Spectroscopy |
Head: Miroslava Novotná (miroslava.novotna@vscht.cz)
Phone: +420 220 444 137
building A, room 310b, 3rd floor |
| Laboratory of Mass Spectrometry (MS) |
Head: Josef Chudoba (josef.chudoba@vscht.cz)
Phone: +420 220 444 3812
building A, room S08, ground-floor |
| Laboratory of X-ray Diffractometry and Spectrometry |
Head: Jaroslav Maixner (jaroslav.maixner@vscht.cz)
Phone: +420 220 444 201 or 5023
GSM: +420 730 809 852
building A, room P03 (4th floor), S38 (ground-floor) |
| Laboratory of Thermal-Gravimetric Analysis (TGA) |
Head: Jakub Havlín (jakub.havlin@vscht.cz)
Phone: +420 220 443 834
building B, room S28a, ground-floor |
| Laboratory of Atomic Absorption Spectroscopy (AAS) |
Atomic Absorption Spectroscopy
Head: Dana Pokorná (dana.pokorna@vscht.cz)
Phone: +420 220 443 813
building A, room 406 |
| Laboratory of Organic Elemental Analysis |
Head: Anna Bradíková (anna.bruthansova@vscht.cz)
Phone: +420 220 443 810
building A, room 275, 2nd floor |
| Laboratory of Surface Analysis |
Head: Petr Sajdl (petr.sajdl@vscht.cz)
Phone: +420 220 443 073
building B, room S12, ground-floor |
| Laboratory of transmission electron microscopy |
|
Head: Alena Michalcová (alena.michalcova@vscht.cz)
Phone: +420 220 444 202
building A, room 49, ground-floor
|
[iduzel] => 21615
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /home
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27384] => stdClass Object
(
[nazev] => Laboratory of Thermal-Gravimetric Analysis (TGA)
[seo_title] => Laboratory of Thermal-Gravimetric Analysis (TGA)
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
About thermal analysis
The methods of thermal analysis enable us to offer the following types of sample/material testing:
- Processes taking place under the heating stress connected with mass change (drying, dehydration, oxidation, …)
- Heat exchange (melting, crystallization, glass transition,…)
- Influence of conditions (heating rate, temperature range, atmosphere, …) on thermally-stressed materials
- Thermal stability (decomposition, oxidation, …)
- Design and verification of heat treatment procedures for a wide range of materials
Principles
Thermogravimetric analysis (TGA)
A sample is subjected to a controlled heating/cooling program (non-isothermal or isothermal) and its weight is measured over time at a range of temperatures.
Differential thermal analysis (DTA)
The sample is heated/cooled at the chosen temperature rate at the same time as a reference, which will remain constant. The temperature difference caused by changes in the sample is recorded and the type of interaction monitored as it takes place. The dependence of the temperature difference on both temperature and time shows the heat aspect of the interaction taking place during the heating or cooling of the sample (exothermic or endothermic effects).
Differential scanning calorimetry (DSC)
DSC monitors heat effects associated with the phase transitions and chemical reactions as a function of temperature. Through appropriate calibration, the results are converted from microvolts to milliwatts. These results show the effects of the interactions and amount of exchanged energy.
Mass spectrometry (MS) combined with thermal analysis (TA)
The quadrupole mass spectrometry analyser enables the detection of chosen masses up to 300 amu. The masses are related to the gases that evolve during the thermal treatment of the sample.
Fourier transform infrared spectroscopy (FTIR) combined with thermogravimetric analysis (TGA)
The infrared spectrometer enables the analysis of the gases that evolve from the thermal analyser during sample analysis.
[iduzel] => 27384
[canonical_url] => //clab.vscht.cz/tga-en
[skupina_www] => Array
(
)
[url] => /tga-en
[sablona] => stdClass Object
(
[class] => stranka_submenu
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27383] => stdClass Object
(
[nazev] => Laboratory of X-ray Diffractometry and Spectrometry
[seo_title] => Laboratory of X-ray Diffractometry and Spectrometry
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
Laboratory of X-ray diffractometry and spectrometry is part of the Central Laboratories at the University of Chemical Technology in Prague. Its main mission is the determination of phase composition using X-ray diffraction analysis(XRD) involving the measurement and evaluation of diffraction patterns and elemental composition by X-ray fluorescence analysis (XRF) of samples supplied from university departments but also from outside the university. In addition to the basic service, specialized or complex experiments, including evaluation can be performed.
X-ray powder diffraction analysis (XRPD, mineralogical analysis, phase analysis) could be used to measure solid samples in the form of powders or flat compacts to determine amorphous or crystalline state of samples. The determination of crystalline phases is performed on the base of comparison measured d, I values with d,I values in databases PDF-4+(Powder diffraction file, inorganic measured and calculated data) or PDF-4/Organics (organic calculated data). Database PDF-4+ includes cards (reference 04-) with x,y,z coordinates of atoms, which could be used for quantitative Rietveld`s analysis.
XRD analysis is mainly used to characterise solid samples, inorganic and organic. Laboratory deals with all chemical problems having relation to solid phase, solid state reactions and heterogeneous systems. It can provide information about progress of solid state reaction, about qualitative and quantitative phase composition of solids, crystalline polymorphism, crystallite size (1-500nm), degree of crystallinity, structural perfection, texture. Special area is molecular and crystal structure of organic or inorganic compounds from single crystals, high-temperature measurement of powders in the range 20-1400°C.
XRD became an indispensable method for studies of corrosion processes, catalytic reactions, synthesis of ceramic materials and pharmaceutic substances(API), research of semiconductors. XRD is not a trace analysis, 0.1 hm.%. is the Limit of Detection(LOD). Recommended quantity of sample for XRD is in section Service. XRD measurement is non-destructive and sample can be used for another analysis.
MicroXRD deals with spot analysis in range 0.1-2 mm. The main advantage is possibility to analyse no-plane compacts in different shapes (e.g. pipes, wires).
X-Ray fluorescent analysis is used to determine elements in range Be-U in powders or compact samples. It is possible to measure liquids as well with restriction to range F-U and worse detection limits for F-Si. Liquids are measured in cups, bottom of the cup is foil, usually 6 micrometer mylar or 4 micrometer polypropylene. Measured liquid must not react with foil, preferably having pH in range 5-7(F-Mg are measured with WD spectrometers). Thera are almost no restriction on pH and reactivity of liquid when ED spectrometer is used, but only Al-U elements can be measured. XRF is suitable for determination of more elements simultaneously (standard F-U), limit of detection generally improves with growing atomic (proton) number, advantage is low overlap among elements, light elements Be-Si should be measured on samples with smooth surface. This is really necessary for Be-O. Recommended quantity of sample for XRD is in section Service. The measurement is mostly non-destructive (warning-glass may darkened). The element composition of glass, cements, ashes, clays, alloys is the main application.
X-ray single crystals diffraction is used for determination of molecular and crystal structure of organic or inorganic moleculs(up to 100 nonH atoms in asymmetric part of cell). Single crystal with size in range 100-1000microns is necessary. Growing of single crystals from solutions can be performed in laboratory, at least 50mg of sample and knowledge about solubility in different solvents is necessary.
[urlnadstranka] =>
[iduzel] => 27383
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /rtg-en
[sablona] => stdClass Object
(
[class] => stranka_submenu
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27382] => stdClass Object
(
[nazev] => Laboratory of Infra-Red (IR) and Raman Spectroscopy
[seo_title] => Laboratory of Infra-Red (IR) and Raman Spectroscopy
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
Laboratory of Molecular Spectroscopy is the part of the Central Laboratories of the University of Chemistry and Technology (UCT) in Prague. We are mainly focused on analysis of unknown substances, identifying their functional groups, verifying the purity of substances and determination of mixtures composition using Infrared (FT-IR) and Raman spectroscopy.
Infrared and Raman Spectrometry can be used in many fields, such as structural analysis of organic and inorganic materials, analysis of foreign substances in the environment, analysis of combustion gases, polymers, sorbents, fillers and adhesives of paper and resins, adhesives for denture, analysis of dyes, fillers and emails used in art and historical works.
Infrared Spectrometry is also used in food analysis (determination of sugars or ethanol in alcoholic drinks, analysis of wines and honey), in medicine (bladder stones), in ecology (determination of petrochemical substances in air and soil, determination of respirable silica in the air, detection of allergenic pollen) and in industrial analysis (determination of additives in oils).
The scientific activity of the laboratory is focused on the development and improvement of measurement methods and on combining of the results obtained by various measurement techniques, which lead to a more reliable solution of the analytical problem.
[urlnadstranka] =>
[iduzel] => 27382
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /ir-en
[sablona] => stdClass Object
(
[class] => stranka_submenu
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27381] => stdClass Object
(
[nazev] => Laboratory of Surface Analysis
[seo_title] => Laboratory of Surface Analysis
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
To measure the surface the laboratory is equipped with ESCAProbeP manufactured by Omicron Nanotechnology Ltd. The device is equipped with a monochromator , the two types of ion guns , electron detection with 5 channeltrony , possibility of compensation charging of the sample using a source of low-energy electrons, the source of UV radiation for analysis of valence states , fokusovatelným electron source and a secondary electron detector .
Most frequent analyses
- Oxidation levels of catalysers
- Surface of organic materials (mainly after some processing) and monitoring of surface changes
- Corrosion
- Layers for chemical sensors
- Materials for electronics with measurement of concentration profiles
[iduzel] => 27381
[canonical_url] => //clab.vscht.cz/lap-en
[skupina_www] => Array
(
)
[url] => /lap-en
[sablona] => stdClass Object
(
[class] => stranka_submenu
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[25101] => stdClass Object
(
[nazev] => Laboratory of Organic Elemental Analysis
[seo_title] => Laboratory of Organic Elemental Analysis
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
The elemental analyses are focused on the determination of following elements – carbon, hydrogen, nitrogen, sulfur, chlorine, bromine, iodine and phosphorus. Analytical methods are based on a sample high-temperature burning in oxygen atmosphere and with a subsequent detection of selected burning products.
Solid or liquid samples can be analysed, e.g. pure chemicals, fuels, coal, heavy oils, plant materials, wood, cellulose etc. Some soils, sands, clays, sediments and other samples being partially or predominantly inorganic can be analyzed. The samples are always destroyed by burning (analyses are destructive).
Analyses
1. Simultaneous determination of carbon, hydrogen, nitrogen and sulfur from one sample
Vario EL Cube analyzer is used for this determination. The analyzer is equipped with a TCD detector and additionally IR detector which is used for determination of the low sulfur content ( below 100 ppm).
Simultaneous C, H, N, S determination is based on high-temperature (up to 1200°C) combustion of the sample in the oxygen stream. Gaseous products of combustion (N2, CO2, H2O a SO2) are purified, separated and finally determined by TCD. Typical samples are organic chemicals but lot of inorganic matters can be analyzed as well.
If your sample contains fluorine in any form and quantity, then the sulfur content cannot be determined. Fluorine containing samples can be analyzed only by the Vario EL III analyzer. In the case of flourinated samples, we can only do the determination of carbon, hydrogen and nitrogen content.
2. Trace and semi-trace determination of halogens and sulfur
We are able to determine even very low content of halogens (not fluorine) and sulfur. These analyses are carried out by the Mitsubishi TOX – 100 analyzer.
Trace and semi-trace determination of halogens:
The sample is burnt in the oxygen-argon atmosphere. Hydrogen chloride produced by sample burning flows into the titration cell where the microcoulometric titration by Ag+ ions takes place. Ag+ ions are automatically generated by Ag - electrode.
Trace and semi-trace determination of sulfur:
The sample is burnt in the oxygen-argon atmosphere. Sulfur dioxide produced by sample burning flows into the titration cell where the microcoulometric titration by I3- ions takes place.
Trace determination of sulfur is disturbed by nitrogen and halogens. If the determination has to be done, your samples cannot contain nitrogen and halogens.
Trace determination of halogens (not fluorine) is disturbed by high sulfur and nitrogen content. If the determination has to be done, your samples can contain sulfur and nitrogen up to 10 %wt. In the case of samples of unknown chemical composition, we have to do the simultaneous C, H, N, S determination at first.
3. Determination of chlorine-, bromine- and iodine content
Methods of classical argentometry are used for the determination of the halogens content. However methods are slightly modified to be able to analyse low quantity of halogens. The sample weight needed for the analysis is depended on the expected halogen content.
The sample is burnt in the oxygen atmosphere inside of the Erlenmeyer flask. Gaseous products of burning are absorbed into the working solution of hydrogen peroxide that is subsequently transfered into the titration flask. The halogen content is determined by potentiometric titration using silver nitrate as titrant.
Your sample has to be a solid and easy burnable matter.
If the estimated halogen content is bellow 5 wt% then the sample mass needed for one analysis is at least 100 mg. If the estimated halogen content is above 5 wt% then the sample amount needed for one analysis is 20 mg.
4. Phosphorus determination
The sample is burnt in the oxygen atmosphere and mineralized. The phosphorus content is determined by a modified complexometric titration.
The sample must not contain alkali earth metals.
If the estimated phosphorus content is low (approx. 0.1 wt%) then the sample amount must be at least 800 mg in one analysis!
[iduzel] => 25101
[canonical_url] => //clab.vscht.cz/oea-en
[skupina_www] => Array
(
)
[url] => /oea-en
[sablona] => stdClass Object
(
[class] => stranka_submenu
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[25094] => stdClass Object
(
[nazev] => Mass Spectrometry Laboratory
[seo_title] => Mass Spectrometry Laboratory
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
Mass Spectrometry (MS) laboratory, a part of university central laboratories, provides analytical service in mass spectrometry. Mass spectra of organic substances and their mixtures are measured. Separation techniques of gas chromatography (GC) and reverse-phase liquid chromatography (RP-HPLC) are used.
Available ionization techniques
GC/MS, direct insertion probe, batch inlet:
- Electron Ionization (EI+ 70 eV)
- Methan Chemical Ionization (CI)
LC/MS, FIA (Flow Injection Analysis), RP-HPLC
- Elektrospray ionization (ESI) – positive, negative mode
- Atmospheric Pressure Chemical Ionization(APCI) - positive, negative mode
Mass spectra acquisition
- low (unit) rosolution
- high resolution – mass accuracy better than 5 ppm (2 ppm with a lock-mass)
- acqusition of MS/MS spectra available (ESI, APCI MSn available)
Mass spectra m/z range
- EI + 70 eV, CI: limited by samples stability and possibility of vaporization (usually up to 1000 Da)
- ESI, APCI: up to 4000 Da (valid for single charged ions)
Analyses
- molecular weight, elemental composition confirmation
- organic mixtures analyses (GC/MS)
- organic mixtures analyses (RP HPLC/MS, RP HPLC/UV-DAD) (consultation required)
- quantitative analyses, pollutants screening in complex matrices (consultation required)
- structure characterization – MS/MS experiments, EI+ 70eV
- GC/MS a LC/MS method development (consultation required)
Samples pre-treatment (consultation required)
- SPME, SPE, MEPS etc.
- thermal desorption (TD) – GC/MS
VOC (Volatile Organic Compounds) analyses in atmosphere or gases , emmited from materials (consultation required)
- Tedlar bag sampling
- thermal desorption tubes (e.g.TENAX) sampling
- solvent desorption tubes (e.g. ORBO, DNPH) sampling
- SUMMA cannister sampling
Analyses results
Mass spectra and additional information are provided in the form of pdf files via e-mail, raw files data are provided on a request. Mass spectra can be provided as weel as Excel or Word files including their metadata and list form.
[iduzel] => 25094
[canonical_url] => //clab.vscht.cz/ms-en
[skupina_www] => Array
(
)
[url] => /ms-en
[sablona] => stdClass Object
(
[class] => stranka_submenu
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[24134] => stdClass Object
(
[obsah] =>
[iduzel] => 24134
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
)
[iduzel] => 20082
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
)
[sablona] => stdClass Object
(
[class] => web
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
[api_suffix] =>
)
DATA
stdClass Object
(
[nazev] => Laboratory of NMR Spectroscopy
[seo_title] => Laboratory of NMR Spectroscopy
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
Laboratory of NMR spectroscopy is the part of the Central Laboratories of the University of Chemistry and Technology (ICT) in Prague. The main goal of our laboratory is to measure service NMR samples delivered not only by various school laboratories but also by out-of-school institutions. Apart from basic services we are able to perform more complex non-standard experiments involving their interpretation and evaluation. At the same time our laboratory deals with scientific and teaching activities that involve close cooperation with other departments of ICT.
NMR spectroscopy can be widely used for characterization of organic compounds. In addition to determination of chemical structure of various products and reactions intermediates, NMR is applicable also for mixture component analysis, kinetic and thermodynamic measurements. NMR is also a powerful tool for quantitative and qualitative calculation of geometry of the molecules and their dynamics.
Enormous development of advanced techniques enabled NMR spectroscopy to become the significant method for calculation of the structure of biomacromolecules – proteins and nucleic acids. At present we are solving the spatial geometry of selected proteins of Mason-Pfizer monkey virus.
[submenuno] =>
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[newurl_domain] => 'clab.vscht.cz'
[newurl_jazyk] => 'en'
[newurl_akce] => '/nmr_en'
[newurl_iduzel] => 27703
[newurl_path] => 1/20076/20078/20082/27703
[newurl_path_link] => Odkaz na newurlCMS
[iduzel] => 27703
[platne_od] => 08.06.2016 12:17:00
[zmeneno_cas] => 08.06.2016 12:19:06.672217
[zmeneno_uzivatel_jmeno] => Jan Prchal
[canonical_url] => //clab.vscht.cz/nmr_en
[idvazba] => 35231
[cms_time] => 1713609472
[skupina_www] => Array
(
)
[slovnik] => Array
(
)
[poduzel] => stdClass Object
(
[27709] => stdClass Object
(
[nazev] =>
[barva_pozadi] => cervena
[uslideru] => false
[text] => Samples
Equipment
Contact+People
Education
Research
[poduzel] => Array
(
)
[iduzel] => 27709
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] => infobox
[html] =>
[css] =>
[js] =>
[autonomni] => 0
)
)
[27769] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[27770] => stdClass Object
(
[nadpis] =>
[popis] =>
[platne_od] =>
[platne_do] =>
[odkaz] => http://clab.vscht.cz/nmr_en
[text_odkazu] =>
[obrazek_pozadi] => y8lPz3f1AwA.jpg
[barva_textu] =>
[iduzel] => 27770
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] => slider
[html] =>
[css] =>
[js] =>
[autonomni] => 0
)
)
)
[iduzel] => 27769
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] => slider
[html] =>
[css] =>
[js] =>
[autonomni] => 0
)
)
[27704] => stdClass Object
(
[nazev] => Laboratory of NMR Spectroscopy
[seo_title] => Samples
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] => Laboratory is equipped for measuring liquid samples. We are able to measure broad range of elements (mainly 1H, 13C, 15N, 19F, 31P).
Samples are measured in deuterated solvents, particularly in CDCl3 , DMSO-d6 or D2O . Therefore, it is necessary to first test the solubility of the substance in a these (non-deuterated ) solvents. The standard volume is 0.5 ml of solution.
For the measurement of 1 H spectra is needed sample concentration of at least 10 mmol / liter , i.e. approx. 1.5 mg of sample ( molecular weight 300 g / mol, volume 0.5 ml) .
For the measurement of 13C spectra is needed sample concentration of at least 20 mmol / l , it is approx. 3.0 mg of sample ( molecular weight 300 g / mol, volume 0.5 ml) .
We require filled Analysis Requirements and Payment Form (pdf) .
Each sample must have unique name, otherwise you risk overwriting your previous measurements!
Measured data can be downloaded from http://ra.vscht.cz/spektra/. Data are stored for 90 days and then removed.
Manually added data (only if you were told, they will be here) can be found here.
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[poduzel] => stdClass Object
(
[27714] => stdClass Object
(
[nazev] =>
[barva_pozadi] => cervena
[uslideru] => false
[text] => Samples
Equipment
Contact+People
Education
Research
[iduzel] => 27714
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] => infobox
[html] =>
[css] =>
[js] =>
[autonomni] => 0
)
)
)
[iduzel] => 27704
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /nmr_en/samples
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27705] => stdClass Object
(
[nazev] => Equipment
[seo_title] => Equipment
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] => Two high-resolution pulse NMR spectrometers are avialable in our laboratory:
Bruker 600 AvanceIII
Triple-resonance digital NMR spectrometer with the following parameters:
- actively shielded superconducting magnet UltraShield™ 14.1 T
- digital console
- orthogonal BOSS II shimming system with 34 gradients
- two probeheads:
- 5 mm triple-resonance cryoprobe TCI (1H observe, 13C and 15N manipulation, deuterium decoupling possible), actively shielded z-gradient
- 5 mm TXI – triple-resonance probehead for inverse experiments (1H observe, 13C and 15N manipulation), actively shielded z-gradient

Bruker Avance III 500 MHz
in the configuration as a two-channel high performance Fourier NMR spectrometer equipped with
- gradient unit in z-axis
- fully digital console Avance III
- Bruker shim system BOSS 3
- three special probeheads
- 5 mm BBFO probehead for direct observation of 1H, 15N-31P (39-162 MHz) and 19F nuclei with decoupling of 1H or 19F nuclei
- inverse 5 mm BBI probehead (1H observe, BB/13C decoupling) with z-gradient
- 10 mm broadband probehead in the standard range (31P - 109Ag), 1H decoupling

[poduzel] => stdClass Object
(
[27711] => stdClass Object
(
[nazev] =>
[barva_pozadi] => cervena
[uslideru] => false
[text] =>
Samples
Equipment
Contact+People
Education
Research
[iduzel] => 27711
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] => infobox
[html] =>
[css] =>
[js] =>
[autonomni] => 0
)
)
)
[iduzel] => 27705
[canonical_url] => //clab.vscht.cz/nmr_en/equipment
[skupina_www] => Array
(
)
[url] => /nmr_en/equipment
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27706] => stdClass Object
(
[nazev] => Education
[seo_title] => Education
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
People from laboratory of NMR spectroscopy teach several specialized courses on UCT Prague. More information can be found on the pages of individual subjects (in Czech).
NMR spektroskopie pro studium přírodních látek (N342010)
Metody určování struktury látek – NMR spektroskopie (N108019)
Strukturní biologie (N320087)
[poduzel] => stdClass Object
(
[27712] => stdClass Object
(
[nazev] =>
[barva_pozadi] => cervena
[uslideru] => false
[text] =>
Samples
Equipment
Contact+People
Education
Research
[iduzel] => 27712
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] => infobox
[html] =>
[css] =>
[js] =>
[autonomni] => 0
)
)
)
[iduzel] => 27706
[canonical_url] => //clab.vscht.cz/nmr_en/education
[skupina_www] => Array
(
)
[url] => /nmr_en/education
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27707] => stdClass Object
(
[nazev] => Laboratory of NMR Spectroscopy
[seo_title] => Contacts
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
Contacts
Laboratory location:
Building A, ground floor, room 42.
Head
Laboratory Staff
Telephone:
+420 220 443 805
+420 220 443 840
+420 220 444 065
Contact Adress:
VŠCHT Praha - Centrální laboratoře
Laboratoř NMR spektroskopie
Technická 5
166 28 Praha 6
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[poduzel] => stdClass Object
(
[27721] => stdClass Object
(
[nazev] => Ivana Bartošová
[seo_title] => Ivana Bartošová
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
 ivana.bartosova@vscht.cz
ivana.bartosova@vscht.cz
Telephone
3805 (tel. 220 443 805)
4065 (tel. 220 444 065)
[iduzel] => 27721
[canonical_url] => //clab.vscht.cz/nmr_en/contacts/bartosova
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts/bartosova
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[68439] => stdClass Object
(
[nazev] => Ing. Jan Sýkora, Ph.D.
[seo_title] => Ing. Jan Sýkora, Ph.D.
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
Phone: 3805 (tel. 220 443 805)
e-mail:sykorab@vscht.cz
ORCID
Education and Qualification:
1998 MSc. in chemistry of silicates, University of Chemistry and Technology in Prague
2002 Ph.D. in inorganic chemistry, University of Chemistry and Technology in Prague
2000 ERASMUS stay, Institute Europeen des Membranes, Montpellier, France
Employment
2001 – 2022 senior researcher, Department of Analytical Chemistry, Institute of Chemical Process Fundamentals of the CAS
2006 visiting professor at Institute of Inorganic Chemistry, Graz Technical University, Austria
2023 senior researcher, Laboratory of NMR spectroscopy, UCT Prague
Research Interests:
Development of analytical procedures towards identification of organic compounds in complex matrices. Analysis of synthetic and natural samples via heteronuclear NMR spectroscopy and bio-fluids analysis by omix approaches. Analysis of intermolecular interactions in the solid state and analysis of various types of X-ray diffraction data as a hobby.
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[iduzel] => 68439
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts/sykora
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[68069] => stdClass Object
(
[nazev] => Ing. Radim Novotný
[seo_title] => Ing. Radim Novotný
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
radim.novotny@vscht.cz
5173 (220 445 173)
ORCID
Education and qualifications
2018-
Ph.D. degree in Biochemistry, Faculty of Food and Biochemical Technology, UCT Prague
2017-
Research worker at the NMR laboratory at the UCT Prague
2016-2018
Master's degree in General and Applied Biochemistry, Faculty of Food and Biochemical Technology, UCT Prague (topic: Identification of Novel Interaction Partners of Mason-Pfizer Monkey Virus Matrix Protein)
2012-2016
Bachelor's degree in Biochemistry and Biotechnology, Faculty of Food and Biochemical Technology, UCT Prague (topic: Cytocompatibility of metal alloys for temporary orthopaedic application)
Current Research Topics in NIVB
Study of structures and interactions of capsid proteins of selected flaviviruses with parts of viral genomic RNA and endoplasmic reticulum membrane. Preferential focus on the capsid protein of tick-borne encephalitis virus.
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[iduzel] => 68069
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts/novotny
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27722] => stdClass Object
(
[nazev] => Ing. Lukáš Vrzal
[seo_title] => Ing. Lukáš Vrzal
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
lukas.vrzal@vscht.cz
3805 (tel. 220 443 805)
Education and qualifications
2014
Ph.D. student, Faculty of Food and Biochemical Technology, UCT Prague
(topic: Fragment-based drug design.)
2012-2014
Master’s degree in Synthesis and Production of Drugs, Faculty of Chemical Engineering, UCT Prague
(topic: Application of alignment media in structural analysis of calix[4]arenes.)
2009-2012
Bachelor’s degree in Chemistry, Faculty of Chemical Engineering, UCT Prague
(topic: Study of the spatial structure of isomers of bridged chiral calix[4]arene using residual dipolar coupling constants.)
Research interests
Structural study of small organic compounds (calix[n]arene derivatives) via NMR spectroscopy, especially using residual dipolar coupling constants (RDCs).
Fragment-based drug discovery using Saturation transfer difference (STD) and Water LOGSY (WL) techniques.
Publications
Vrzal L., Flídrová K., Tobrman, T., Dvořáková, H., Lhoták, P., Use of residual dipolar couplings in conformational analysis of meta-disubstituted calix[4]arenes. (2014) Chem. Com., 50(57), 7590-7592.
doi:10.1039/C4CC02274B
Váňa L.; Vrzal L., Dvořáková H., Himl M., Linhart I. Direct arylation of adenine by fluoro- and chloronitrobenzenes. Effect of microwaves. (2013) Synth. Com., 44(6), 788-799.
doi:10.1080/00397911.2013.831902
Holub J., Eigner V., Vrzal L., Dvořáková H., Lhoták P. Calix[4]arenes with intramolecularly bridged meta positions prepared via Pd-catalysed double C-H activation. (2013) Chem. Com., 49(27),2798-2800.
doi:10.1039/c3cc40655e
[iduzel] => 27722
[canonical_url] => //clab.vscht.cz/nmr_en/contacts/vrzal
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts/vrzal
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27717] => stdClass Object
(
[nazev] => Ing. Hana Dvořáková, CSc.
[seo_title] => Ing. Hana Dvořáková, CSc.
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
hana.dvorakova@vscht.cz
3840 (tel. 220 443 840)
Education and Qualification.
1996- Research Assistant at the NMR laboratory, UCT Prague
1994 One year stay at the Organic Chemistry Department of the University of Leicester (Great Britain)
1991 Ph.D. in organic chemistry, Institute of Organic Chemistry and Biochemistry, Academy of Sciences of the Czech Republic, Prague. Thesis: Acyclic Analogues of Nucleosides and Nucleotides with Modified Purine Bases
1983-1995 Research Assistant at the Institute of Organic Chemistry and Biochemistry, Academy of Sciences of the Czech Republic, Prague
1978 MSc. in organic chemistry, UCT Prague (theme: Electrochemical oxidations of bezofuranes)
Research interests
Application of high resolution NMR spectroscopy in the field of calixarenes, resorcinarenes and natural compounds. Studies of conformational a dynamic properties of molecules; kinetics and thermodynamics of chemical reactions.
Selected publications
Krovacek M., Dvorakova H., Votruba I., Cisarova I., Dvorak D. (2012) 6-Alkynylpurines bearing electron acceptor substituents: preparation, reactivity in cycloaddition reactions and cytostatic activity. Collection of Czechoslovak Chemical Communications, 76(12), 1487-1507. doi:10.1135/cccc2011176
Kundrat O., Dvorakova H., Bohm S., Eigner V., Lhotak P. (2012) S-Alkylation of Thiacalixarenes: How the Regio- and Stereoselectivities Depend on the Starting Conformation. Journal of Organic Chemistry, 77(5), 2272-2278. doi:10.1021/jo202571h
Kaminsky J., Dvorakova H., Stursa J., Moravcova J. (2011) Collection of Czechoslovak Chemical Communications, 76(10), 1199-1222. doi:10.1135/cccc2010104
Menova P., Eigner V., Cejka J., Dvorakova H., Sanda M., Cibulka R. (2011) Synthesis and structural studies of flavin and alloxazine adducts with O-nucleophiles. Journal of Molecular Structure, 1004(1-3), 178-187. doi:10.1016/j.molstruc.2011.08.002
Kundrat O., Eigner V., Dvorakova H., Lhotak P. (2011) S-Alkylation of Thiacalixarenes: A Long-Neglected Possibility in the Calixarene Family. Organic Letters, 13(15), 4032-4035. doi:10.1021/ol201546y
Menova P., Kafka F., Dvorakova H., Gunnoo S., Sanda M., Cibulka R. (2011) Pyrazinium Salts as Efficient Organocatalysts of Mild Oxidations with Hydrogen Peroxide. Advanced Synthesis & Catalysis. 353(6), 865-870. doi:10.1002/adsc.201000906
Jurok R., Cibulka R., Dvorakova H., Hampl F., Hodacova J. (2010) Planar Chiral Flavinium Salts - Prospective Catalysts for Enantioselective Sulfoxidation Reactions. European Journal of Organic Chemistry. (27), 5217-5224 doi:10.1002/ejoc.201000592
Chlubnova I., Filipp D., Spiwok V., Dvorakova H., Daniellou R., Nugier-Chauvin Caroline,. Kralova B., Ferrieres V. (2010) Enzymatic synthesis of oligo-D-galactofuranosides and L-arabinofuranosides: from molecular dynamics to immunological assays. Organic & Biomolecular Chemistry. 8(9), 2092-2102. doi:10.1039/b926988f
Dolensky B., Kvicala J., Paleta O., Lang J., Dvorakova H., Cejka J. (2010) Trifluoromethylated (tetrahydropyrrolo) quinazolinones by a new three-component reaction and facile assignment of the regio- and stereoisomers formed by NMR spectroscopy. Magnetic Resonance in Chemistry, 48(5), 375-385. doi:10.1002/mrc.2580
Benesova E., Lipovova P., Dvorakova H., Kralova B. (2010) ß-d-Galactosidase from Paenibacillus thiaminolyticus catalyzing transfucosylation reactions. Glycobiology, 20(4), 442-451. doi:10.1093/glycob/cwp196
Hornicek J., Dvorakova H., Bour P. (2010) Intramolecular Proton Transfer in Calixphyrin Derivatives. Journal of Physical Chemistry A, 114(10), 3649-3654. doi:10.1021/jp911598w
Zurek J., Cibulka R., Dvorakova H., Svoboda J. (2010) N 1,N 10-Ethylene-bridged flavinium salts derived from -valinol: synthesis and catalytic activity in H2O2 oxidations. Tetrahedron Letters, 51(7), 1083-1086. doi:10.1016/j.tetlet.2009.12.096
Kundrat O., Dvorakova H., Eigner V., Lhotak P. (2010) Uncommon Regioselectivity in the Thiacalix[4]arene Series: Gross Formylation of the Cone Conformer. Journal of Organic Chemistry, 75(2), 407-411. doi:10.1021/jo902240h
Kundrat, O., Dvorakova, H., Cisarova, I., Pojarova, M., Lhotak, P. (2009) Org. Lett. 11, 4188-4191. doi:10.1021/ol901812m
Keder, R., Dvorakova, H., Dvorak, D. (2009) Eur. J. Org. Chem. 1522-1531. doi:10.1002/ejoc.200801002
Simanova, M., Dvorakova, H., Stibor, I., Pojarova, M., Lhotak, P. (2008) Tetrahedron Lett. 49, 1026-1029. doi:10.1016/j.tetlet.2007.12.004
Dvorakova, H., Lang, J., Vlach, J., Sykora, J., Cajan, M., Himl, M., Pojarova, M., Stibor, I., Lhotak, P. (2007) J. Org. Chem. 72, 7157-7166. doi:10.1021/jo070927i
Tobrman, T., Meca, L., Dvorakova, H., Cerny, J., Dvorak, D. (2006) Organometallics 25, 5540-5548. doi:10.1021/om0605837
Dvorakova, H., Stursa, J., Cajan, M., Moravcova, J. (2006) Eur. J. Org. Chem. 4519-4527. doi:10.1002/ejoc.200600386
Bernatkova, M., Dvorakova, H., Andrioletti, B., Kral, V., Bour, P. (2005) J. Phys. Chem. A 109, 5518-5526. doi:10.1021/jp050746p
Dolensky, B., Kroulik, J., Kral, V., Sessler, J. L., Dvorakova, H., Bour, P., Bernatkova, M., Bucher, C., Lynch, V. (2004) J. Am. Chem. Soc. 126, 13714-13722. doi:10.1021/ja048075g
Novak, J., Linhart, I., Dvorakova, H. (2004) Eur. J. Org. Chem. 2738-2746. doi:10.1002/ejoc.200300811
Stastny, V., Stibor, I., Dvorakova, H., Lhotak, P. (2004) Tetrahedron 60, 3383-3391. doi:10.1016/j.tet.2004.02.036
Stursa, J., Dvorakova, H., Smidrkal, J., Petrickova, H., Moravcova, J. (2004) Tetrahedron Lett. 45, 2043-2046. doi:10.1016/j.tetlet.2004.01.080
Lhotak, P., Himl, M., Stibor, I., Sykora, J., Dvorakova, H., Lang, J., Petrickova, H. (2003) Tetrahedron 59, 7581-7585. doi:10.1016/S0040-4020(03)01171-2
Hocek, M. Dvorakova, H. (2003) J. Org. Chem. 68, 5773-5776. doi:10.1021/jo034351i
Dvorakova, H., Masojidkova, M., Holy, A., Balzarini, J., Andrei, G., Snoeck, R., DeClercq, E. (1996) J. Med. Chem. 39, 3263-3268. doi:
Dvorakova, H., Dvorak, D., Holy, A. (1996) Tetrahedron Lett. 37, 1285-1288.
[iduzel] => 27717
[canonical_url] => //clab.vscht.cz/nmr_en/contacts/dvorakova
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts/dvorakova
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27720] => stdClass Object
(
[nazev] => Ing. Tomáš Kroupa
[seo_title] => Ing. Tomáš Kroupa
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
tomas.kroupa@vscht.cz
3840 (tel. 220 443 840)
5173 (tel. 220 445 173)
Education
2011-2016
PhD studies, Biochemistry, UCT Prague (Dissertation thesis: Study of the interaction of immature retroviral particles with cell membranes)
2009-2011
Master studies, Pharmaceutical biotechnology, UCT Prague (Diploma thesis: Effect of potential inhibitors of microbial adhesion on the cell surface hydrophobicity)
Field of research
Molecular virology (structure and function of membrane-associated retroviral proteins).
Selected publications
Prchal, J., Kroupa, T., Ruml, T., Hrabal, R. (2014) Interaction of Mason-Pfizer monkey virus matrix protein with plasma membrane. Front. Microbiol. 4(423), doi: 10.3389/fmicb.2013.00423
Kroupa, T., Prchal, J., Doležal, M., Ruml, T., & Hrabal, R. (2014). Cost-effective method for the preparation of uniformly labeled myristoylated proteins for NMR measurements. Protein Expr. Purif., 99, 6-9. doi:10.1016/j.pep.2014.03.005
[iduzel] => 27720
[canonical_url] => //clab.vscht.cz/nmr_en/contacts/kroupa
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts/kroupa
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27719] => stdClass Object
(
[nazev] => doc. RNDr. Jan Lang, PhD.
[seo_title] => doc. RNDr. Jan Lang, PhD.
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
jan.lang@vscht.cz
22191 2889 (MFF UK)
Education and Qualification
1984–1994 Mgr. studies in the field of Physics of Molecular and Biological Systems, curriculum biophysics at the Faculty of Mathematics and Physics, Charles University in Prague, Czech Republic.
1993–1994 Stay at the Division of Physical Chemistry of the University of Stockholm, Stockholm, Sweden.
1994 Graduated at the Charles University with Magister degree.
1999 Ph.D. in physics of molecular and biological systems at Charles University.
2000 Post-doctoral fellowship with Prof. J. Kowalewski, Stockholm University, Sweden.
since 1997 Research assistant in the NMR laboratory, UCT Prague.
Research interests
Application of high resolution NMR spectroscopy for determination of structural and dynamic properties of small and medium organic and biological molecules. Studies of nuclear relaxation and exchange processes.
Selected publications
Lhoták P., Kaplánek L., Stibor I., Lang J., Dvořáková H; Hrabal R., Sýkora J.: NMR and X-ray analysis of 25,27-dimethoxythiacalix[4] arene: unique infinite channels in the solid state. Tetrahedron Lett. 41, 9339-9344 (2000).
Lang J., Vlach J., Dvořáková H., Lhoták P., Himl M., Hrabal R., Stibor I.: Thermal isomerisation of 25,26,27,28-tetrapropoxy-2,8,14,20-tetrathiacalix[4]arene: Isolation of all four conformers. J. Chem. Soc., Perkin Trans 2, 4, 576
[iduzel] => 27719
[canonical_url] => //clab.vscht.cz/nmr_en/contacts/lang
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts/lang
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27718] => stdClass Object
(
[nazev] => Ing. Jan Prchal, PhD.
[seo_title] => Ing. Jan Prchal, PhD.
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
jan.prchal@vscht.cz
3840 (tel. 220 443 840)
5173 (tel. 220 445 173)
Researcher ID
ORCID
Education:
2013 PhD. in biochemistry UCT Prague. Thesis: Study of M-PMV Matrix Protein Interaction with Cytoplasmic Membrane
2007 Master degree in biochemistry UCT Prague. Study of Structure of T41I/T78I Matrix Protein Mutant of M-PMV
Employment
2014 Short research stay in Dr. Delphine Muriaux’s laboratory, CPBS, CMRS, Montpellier, Francie
2010- Research worker at the NMR laboratory at the UCT Prague
Research Interests:
Virology, study of protein structure and function using NMR spectroscopy
Selected Publications
Vlach J., Srb P., Prchal J., Grocký M., Lang J., Ruml T., Hrabal R. (2009) Nonmyristoylated Matrix Protein from the Mason-Pfizer Monkey Virus Forms Oligomers. J. Mol. Biol., 390(5), 967–980. doi:10.1016/j.jmb.2009.05.063
Srb P., Vlach J., Prchal J., Grocký M., Ruml T., Lang J., Hrabal R. (2011) Oligomerization of a retroviral matrix protein is facilitated by backbone flexibility on nanosecond time scale. J. Phys. Chem. B, 115(11), 2634-2644.doi: 10.1021/jp110420m
Prchal J., Junková P., Strmisková M., Lipov J., Hynek R., Ruml T., Hrabal R. (2011) Expression and purification of myristoylated matrix protein of Mason-Pfizer monkey virus for NMR and MS measurements. Protein Expr. Purif., 79(1), 122-127. doi: 10.1016/j.pep.2011.05.010
Prchal, J., Srb, P., Hunter, E., Ruml, T., Hrabal, R. (2012)The Structure of Myristoylated Mason-Pfizer Monkey Virus Matrix Protein and the Role of Phosphatidylinositol-(4,5)-bisphosphate in its Membrane Binding. J.Mol.Biol., 423(3), 427-438. doi:10.1016/j.jmb.2012.07.021
Junkova, P., Vermachova, M., Prchal, J., Kuckova, S., Hrabal, R., Hynek, R. (2013) Improved Approach for the Labeling of Argininne, Glutamic, and Aspartic Acid Side Chains in Proteins Using Chromatographic Techniques. J. Liq. Chromatogr. Relat. Technol., 36(9), 1221-1230. doi:10.1080/10826076.2012.685918
Rozbeský, D., Krejzová, J., Křenek, K., Prchal, J., Hrabal, R., Kožíšek, M., Weignerová, L., Fiore, M., Dumy, P., Křen, V., Renaudet, O. (2014) Re-Evaluation of Binding Properties of Recombinant Lymphocyte Receptors NKR-P1A and CD69 to Chemically Synthesized Glycans and Peptides. Int. J. Mol. Sci., 15(1), 1271-1283. doi:10.3390/ijms15011271
Prchal, J., Kroupa, T., Ruml, T., Hrabal, R. (2014) Interaction of Mason-Pfizer monkey virus matrix protein with plasma membrane. Front. Microbiol. 4(423), doi: 10.3389/fmicb.2013.00423
Kroupa, T., Prchal, J., Doležal, M., Ruml, T., & Hrabal, R. (2014). Cost-effective method for the preparation of uniformly labeled myristoylated proteins for NMR measurements. Protein Expr. Purif., 99, 6-9. doi:10.1016/j.pep.2014.03.005
Junková, P., Prchal, J., Spiwok, V., Pleskot, R., Kadlec, J., Krásný, L., Hynek, R., Hrabal, R. and Ruml, T. (2016), Molecular aspects of the interaction between Mason—Pfizer monkey virus matrix protein and artificial phospholipid membrane. Proteins, 84: 1717–1727. doi:10.1002/prot.25156
Kerviel A, Dash S, Moncorgé O, Panthu B, Prchal J, Décimo D, et al. (2016) Involvement of an Arginine Triplet in M1 Matrix Protein Interaction with Membranes and in M1 Recruitment into Virus-Like Particles of the Influenza A(H1N1)pdm09 Virus. PLoS ONE 11(11): e0165421. doi:10.1371/journal.pone.0165421
Kroupa T., Langerová H., Doležal M., Prchal J., Spiwok V., Hunter E., Rumlová M., Ruml T., Hrabal R. (2016), Membrane Interactions of the Mason-Pfizer Monkey Virus Matrix Protein and Its Budding Deficient Mutants. J.Mol.Biol. 428(23):4708-4722. doi: 10.1016/j.jmb.2016.10.010.
JUNKOVA, P.; PLESKOT, R.; PRCHAL, J.; SYS, J. et al. Differences and commonalities in plasma membrane recruitment of the two morphogenetically distinct retroviruses HIV-1 and MMTV (vol 295, pg 8819, 2020). Journal of Biological Chemistry, 295, n. 31, p. 10869-10869, Jul 2020. DOI: 10.1074/JBC.AAC120.015073
PRCHAL, J.; SYS, J.; JUNKOVA, P.; LIPOV, J. et al. Interaction Interface of Mason-Pfizer Monkey Virus Matrix and Envelope Proteins. Journal of Virology, 94, n. 20, Oct 2020. DOI:10.1128/JVI.01146-20
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[iduzel] => 27718
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts/prchal
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27715] => stdClass Object
(
[nazev] => prof. Ing. Richard Hrabal, CSc.
[seo_title] => prof. Ing. Richard Hrabal, CSc.
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
Head of Laboratory
Phone: 3805 (tel. 220 443 805)
e-mail: richard.hrabal@vscht.cz
Orcid
Education and Qualification:
2017 named professor of biochemistry, UCT Prague
2007 habilitation in biochemistry, UCT Prague
1989 Ph.D. in organic chemistry UCT in Prague. Thesis: Synthesis, structure and reactivity of highly fluorinated dienes and trienes
1988 Postgradual course in liquid and solid phase NMR spectroscopy Technical University in Bratislava
1982 MSc. in organic chemistry Institute of Chemical Technology in Prague. Thesis: Study of fluorinated dienes
Employment
1996- Head of the NMR laboratory at the UCT Prague
1993-1996 Research Associate na Biotechnology Research Institute, div. Biomolecular Interactions. National Research Council of Canada
1992-1993 Head of the NMR laboratory at the UCT Prague
1990 Short term visit at the Inorganic Chemistry Department of the Oxford University Prof. R.J.P. Williams. (Study of binding zinc ion to short peptides)
1987-1992 Research Assistant in the NMR Laboratory at the UCT Prague
1986-1987 Research Assistant at the Department of Organic Chemistry of the UCT Prague
Research Interests:
High resolution NMR spectroscopy and its application in chemistry, biochemistry and molecular biology. Molecular modelling, especially related to the elucidation of structures of petides, proteins and their complexes.
Selected Publications
- Šebek P., Nešpůrek S., Hrabal R., Adamec M., Kuthan J.: Novel Preparation Photochromic Properties of 2,4,4,6-tetraaryl-4H-thiopyrans. J.Chem. Soc.-Perkin Trans 2 1301-1308 (1992) [IF 1.863, cited 34]
- Srinivasan J., Hu S., Hrabal R., Zhu Y., Komives E.A., Ni F.: Thrombin-Bound Structure of an EGF Subdomain from Human Thrombomodulin Determined by Tranferred Nuclear Overhauser Effect. Biochemistry 33, 13553 (1994) [IF 3.922, cited 28]
- Hrabal R., Komives. E.A., Ni F.: Structure Resiliency of an EGF-like Subdomain Bound to its Target Protein, Thrombin. Protein Science, 5, 195 (1996) [IF 3.787, cited 20]
- Lang J., Dvořáková H., Bartošová I., Lhoták P., Stibor I., Hrabal R.: Conformational flexibility of a novel tetraethylether of thiacalix[4]arene. A comparison with the "classical" methylene-bridged compounds. Tetrahedron Lett. 40, 373-376 (1999) (IF 2.615, cited 41]
- Veverka V., Bauerová H., Zábranský A., Lang J., Ruml T., Pichová I., Hrabal R.: Three-dimensional Structure of a Monomeric Form of a Retroviral Protease. J. Mol.Biol. 333, 771-780 (2003) [IF 4.472, cited 5]
- Hrabal R., Chen Z., James S., Bennet H.P.J., Ni F.: The hairpin stack fold, a novel protein architecture for a new family of protein growth factors. Nature Structural Biology, 3, 747-652 (1996) [IF 11.085, cited 35]
- Zábranská H., Tůma R., Kuhl I., Svatoš A., Ruml T., Hrabal R., Pichová I.: The role of the S-S bridge in retroviral protease fiction And virion maturation. J. Mol. Biol. 365, 1493-1502 (2007) [IF 4.472, cited 2]
- Vlach J., Lipov J., Rumlová M., Veverka V., Lang J., Srb P., Knejzlík Z., Pichová I., Hunter E., Hrabal R., Ruml T.: D-retrovirus morhogenetic switch driven by the targeting signal accessibility to Tctex-1 of dynein. Proc. Natl. Acad. Sci. USA 105, 10565-10570 (2008) [IF 9.598, cited 1]
- Vlach J., Srb P., Prchal J., Grocký M., Lang J., Ruml T., Hrabal R. (2009) Nonmyristoylated Matrix Protein from the Mason-Pfizer Monkey Virus Forms Oligomers. J. Mol. Biol., 390(5), 967–980. doi:10.1016/j.jmb.2009.05.063
- Srb P., Vlach J., Prchal J., Grocký M., Ruml T., Lang J., Hrabal R. (2011) Oligomerization of a retroviral matrix protein is facilitated by backbone flexibility on nanosecond time scale. J. Phys. Chem. B, 115(11), 2634-2644.doi: 10.1021/jp110420m
- Prchal, J., Srb, P., Hunter, E., Ruml, T., Hrabal, R. (2012)The Structure of Myristoylated Mason-Pfizer Monkey Virus Matrix Protein and the Role of Phosphatidylinositol-(4,5)-bisphosphate in its Membrane Binding.J.Mol.Biol., in press, doi:10.1016/j.jmb.2012.07.021
- Junková, P., Prchal, J., Spiwok, V., Pleskot, R., Kadlec, J., Krásný, L., Hynek, R., Hrabal, R. and Ruml, T. (2016), Molecular aspects of the interaction between Mason—Pfizer monkey virus matrix protein and artificial phospholipid membrane. Proteins, 84: 1717–1727. doi:10.1002/prot.25156
- Kroupa T., Langerová H., Doležal M., Prchal J., Spiwok V., Hunter E., Rumlová M., Ruml T., Hrabal R. (2016), Membrane Interactions of the Mason-Pfizer Monkey Virus Matrix Protein and Its Budding Deficient Mutants. J.Mol.Biol. 428(23):4708-4722. doi: 10.1016/j.jmb.2016.10.010.
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[iduzel] => 27715
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts/hrabal
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27713] => stdClass Object
(
[nazev] =>
[barva_pozadi] => cervena
[uslideru] => false
[text] =>
Samples
Equipment
Contact+People
Education
Research
[iduzel] => 27713
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] => infobox
[html] =>
[css] =>
[js] =>
[autonomni] => 0
)
)
)
[iduzel] => 27707
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /nmr_en/contacts
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
[27708] => stdClass Object
(
[nazev] => Research
[seo_title] => Research
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
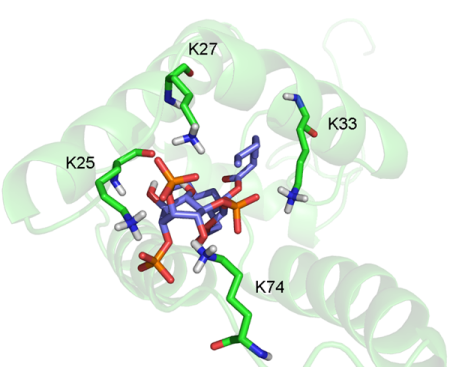
Research in this laboratory can be divided in two branches. The first is focused on small organic molecules (calixarenes, nucleosides, flavines, small natural compounds and their analogues, etc.) and their structure or conformation. The other is focused on biological macromolecules - proteins, mainly on matrix protein of Mason-Pfizer monkey virus, where we focus mainly on the determination of the protein structure and its interaction with other molecules. The latest results can be found in publications listed under the profiles of their autors.
Metabolomics

[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[poduzel] => stdClass Object
(
[27710] => stdClass Object
(
[nazev] =>
[barva_pozadi] => cervena
[uslideru] => false
[text] =>
Samples
Equipment
Contact+People
Education
Research
[iduzel] => 27710
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] => infobox
[html] =>
[css] =>
[js] =>
[autonomni] => 0
)
)
[69645] => stdClass Object
(
[nazev] => Metabolomics
[seo_title] => Metabolomics
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
NMR-omics, compound profiling in 1H NMR spectra
NMR spectroscopy can be also utilized in the analysis of complicated mixtures of organic substances, advantageously. If we intend to identify or even quantify the individual substances in a mixture, we have to know a spectrum of pure substance. The identification of a given substance can be then realized on the basis of a single signal, which is easily recognizable in the spectrum of the whole mixture. Subsequently, it is necessary to find out whether all the remaining signals of a given substance are present in the spectrum. If so, the intensity of all its signals have to be adjusted proportionally to the intensities of the signals in the spectrum of the mixture. Comparing the signal intensity with the signal of a standard, we get the information on compound concentration in the sample. This procedure is called compound profiling and it works best when analyzing a pre-defined group of substances in a given matrix. Such samples can be, for example, bodily fluids, in which we look for individual metabolites, this is called metabolomics. Based on the concentrations of the most common organic substances such as simple organic acids, carbohydrates, alcohols, etc., we can determine the origin and quality of food using so-called foodomics. Aerosolomics, on the other hand, deals with determining of concentrations of substances in the air and subsequently the origin of air pollution. Compound profiling is a universal approach that can find wide application, but it is still a quite new topic, at least in the Czech Republic. Advanced methods of statistical analysis are an integral part of the processing of the obtained data. It enables to find subtle differences in the concentrations between pre-defined groups. We can thus distinguish between samples of healthy individuals and patients, or prediction models can be constructed to predict how an at-risk individual will develop in future. We are currently dealing with projects focused on, for example, early diagnosis of pancreatic cancer, Alzheimer's disease or hepatocellular carcinoma. We mainly work with blood samples, but we have also an experience with breath condensate or cerebrospinal fluid samples. Based on statistical analysis, we can determine metabolites with significant alterations in concentration and subsequently identify affected metabolic pathways. Using NMR metabolomics, the development of the disease can be monitored over time, or the individual's response to ongoing treatment can be studied. Targeted analysis can also monitor the metabolism of a given drug in the body. In short, there are many possible directions of research...
Michálková, L.; Horník, Š.; Sýkora, J.; Habartová, L.; Setnička, V.; Bunganič, B. Early Detection of Pancreatic Cancer in Type 2 Diabetes Mellitus Patients Based on H-1 NMR Metabolomics. J. Proteome Res. 2021, 20(3), 1744–1753. https://doi.org/10.1021/acs.jproteome.0c00990
Horník, Š.; Michálková, L.; Sýkora, J.; Ždímal, V.; Vlčková, S.; Dvořáčková, S.; Pelclová, D. Effects of Workers Exposure to Nanoparticles Studied by NMR Metabolomics. Appl. Sci. 2021, 11, 6601. https://doi.org/10.3390/app11146601
Horník, Š.; Sýkora, J.; Pokorná, P.; Vodička, P.; Schwarz, J.; Ždímal, V. Detailed NMR analysis of water-soluble organic compounds in size-resolved particulate matter seasonally collected at a suburban site in Prague. Atmospheric Environment 2021, 267, 118757. https://doi.org/10.1016/j.atmosenv.2021.118757
Horník, Š.; Sýkora, J.; Schwarz, J.; Ždímal, V. Nuclear Magnetic Resonance Aerosolomics: A Tool for Analysis of Polar Compounds in Atmospheric Aerosols. ACS Omega 2020, 5(36), 22750–22758. https://doi.org/10.1021/acsomega.0c01634
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[iduzel] => 69645
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /nmr_en/research/metabolomika
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
)
[iduzel] => 27708
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /nmr_en/research
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
)
)
[sablona] => stdClass Object
(
[class] => stranka
[html] =>
[css] =>
[js] =>
[autonomni] => 1
)
[api_suffix] =>
)
 [logo_mobile_href] => /
[logo_mobile] =>
[logo_mobile_href] => /
[logo_mobile] => 










